Orthofix Completes Acquisition of FITBONE Limb Lengthening System
Orthofix Completes Acquisition of FITBONE Limb Lengthening System
LEWISVILLE, Texas--(BUSINESS WIRE)--Orthofix Medical Inc. (NASDAQ:OFIX), a global medical device company focused on musculoskeletal healing products, today announced it has completed the acquisition of assets associated with the FITBONE® intramedullary lengthening system for limb lengthening of the femur and tibia bones. The transaction also includes other potential applications of the technology which are in development, including the FITSPINE® system for fusionless surgery to treat early onset scoliosis. With the addition of the FITBONE assets, Orthofix becomes the only orthopedic company that offers a comprehensive portfolio of both internal and external fixation solutions for limb reconstruction.
“The acquisition of the FITBONE® intramedullary lengthening system further demonstrates Orthofix’s commitment to investing in differentiated products that are a strong strategic fit within our core businesses,” said Orthofix President and Chief Executive Officer Jon Serbousek. “Adding this technology to our current limb reconstruction portfolio enables us to offer physicians solutions to meet the needs of their patients which may require internal or external fixation procedures. We look forward to developing other applications of this exciting platform.”
Terms of the agreement executed with Wittenstein SE, a privately-held Germany-based company who developed the technology, include $18 million in cash closing consideration and a manufacturing supply contract with Wittenstein SE.
The FITBONE system will be a part of the Orthofix Extremities portfolio of solutions that include the TL-HEX™ computer-assisted ring fixation system for external limb lengthening and the eight-Plate Guided Growth System™ for correcting angular growth deformities in pediatric patients. To learn more about Orthofix’s dedication to helping surgeons and limb deformity correction patients, please visit JuniOrtho.club.
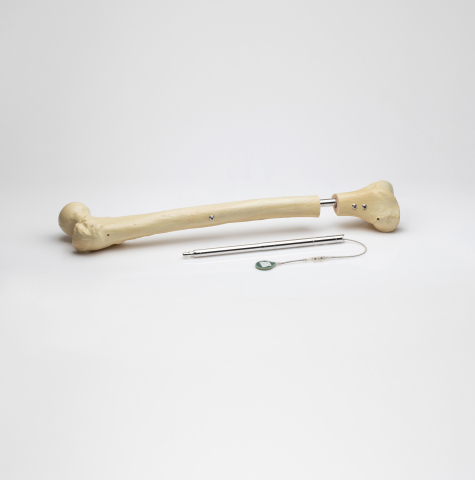
Consisting of an intramedullary lengthening nail that is surgically implanted in the bone through a minimally invasive procedure, the FITBONE system includes an external telemetry control set that manages the distraction process. The patient’s treatment is also supported through the FITBONE app that guides the patient throughout the limb-lengthening treatment. Over 3,500 cases in more than 15 countries have been performed with the FITBONE system.
The FITBONE intramedullary lengthening system is available in the U.S. under a U.S. Food and Drug Administration 510(k) clearance and in European Countries under a CE Mark approval.
About Orthofix
Orthofix Medical Inc. is a global medical device company focused on musculoskeletal products and therapies. The Company’s mission is to improve patients' lives by providing superior reconstruction and regenerative musculoskeletal solutions to physicians worldwide. Headquartered in Lewisville, Texas, Orthofix’s spine and orthopedic extremities products are distributed in more than 70 countries via the Company's sales representatives and distributors. For more information, please visit www.orthofix.com.
Forward Looking Statements
This communication contains certain forward-looking statements under the Private Securities Litigation Reform Act of 1995. These forward-looking statements, which may include, but are not limited to, statements concerning the estimates, projections, financial condition, results of operations and businesses of Orthofix and its subsidiaries, Wittenstein SE and their respective companies’ product portfolios, are based on Orthofix management's current expectations and estimates and involve risks and uncertainties that could cause actual results or outcomes to differ materially from those contemplated by the forward-looking statements.
The forward-looking statements in this release do not constitute guarantees or promises of future performance. Factors that could cause or contribute to such differences may include, but are not limited to risks, including difficulties commercializing products and integrating the product into Orthofix’s business, inaccuracies in Orthofix’s estimates and projections of future product sales, including the current and future size of the worldwide and U.S. limb reconstruction market, FDA and regulatory approval risks, and other risks described in the "Risk Factors" section of our 2019 Annual Report on Form 10-K, as well as in other reports that we file in the future. Existing and prospective investors are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date hereof. The Company undertakes no obligation to update or revise the information contained in this press release.
Contacts
Mark Quick
Investor Relations
Tel 214 937 2924
markquick@orthofix.com
Denise Landry
Media Relations
Tel 214 937 2529
deniselandry@orthofix.com