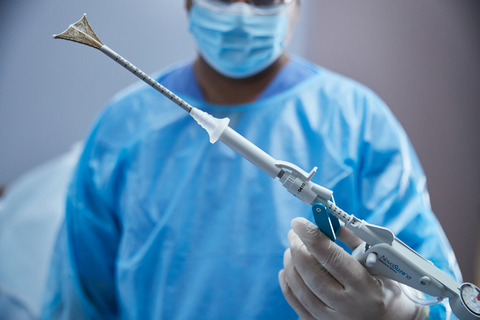
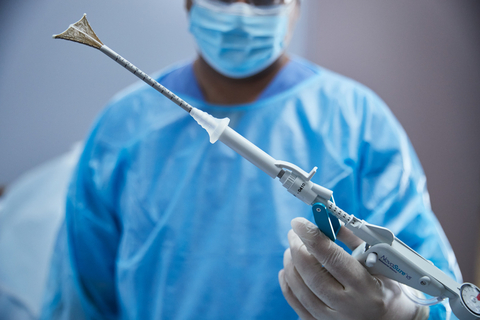
MARLBOROUGH, Mass.--(BUSINESS WIRE)--Hologic Inc. (Nasdaq: HOLX), a global leader in women's health, has announced approval of the NovaSure® V5 global endometrial ablation (GEA) device in Canada and Europe. This innovative new version incorporates enhanced features designed to treat a wide range of cervical and uterine anatomies. It builds on a world-class track record for NovaSure, including 3 million patients who have benefited from the device.
“Since its launch over 20 years ago, we have constantly listened to our customers’ feedback, which has driven continuous design improvements for NovaSure,” said Jan Verstreken, Group President, International at Hologic. “This has enabled us to develop our technology to further meet their needs and those of the women they treat, ultimately delivering better outcomes for these women.”
NovaSure V5 has an updated cervical seal, featuring EndoForm™ technology, designed to increase the sealing surface and accommodate a range of cervical canals and anatomical variability.1,2,3 The device’s AccuSheath™ markings are designed to improve the accuracy and confidence of seating and fundal placement.4 Additionally, NovaSure V5 is equipped with SureClear™ technology, Hologic's unique fluid removal system, which provides integrated suction through the array by constant tissue contact while simultaneously removing ablation byproducts such as vapor and fluid.5
“I welcome the new features on the NovaSure V5. These will support both inexperienced and more established users in performing effective, more efficient and accurate procedures,” said Dr. Philippe Y. Laberge, M.D., FRCSC, ACGE based at the CHU de Québec-Université Laval in Québec, Canada. “The addition of markings to the shaft will enable users to correctly place the device before initiating a procedure. The cervical seal is a significant improvement as there is less chance of leaks, allowing the completion of pre-procedure checks and the procedure itself.”
The NovaSure device enables an endometrial ablation procedure for the treatment of abnormal uterine bleeding, with 97% patient satisfaction and 87% of patients avoiding a hysterectomy at 10 years.6,7
For more information about the benefits and risks of NovaSure V5, visit: www.gynsurgicalsolutions.co.uk/NovaSureV5.
About Hologic
Hologic Inc. is a global medical technology innovator primarily focused on improving women’s health and well-being through early detection and treatment. Its advancements include invention of the world’s first commercial 3D mammography system to find breast cancer; leadership in testing for cervical cancer, sexually transmitted infections, respiratory illnesses and the virus that causes COVID-19; and minimally invasive surgical technologies for uterine fibroids and abnormal uterine bleeding.
The company also champions women through the Hologic Global Women’s Health Index, which provides a science-backed data roadmap for improving women’s well-being, and Project Health Equality, which elevates awareness, research insights and access to quality care for underserved women.
Forward-Looking Statements
This press release may contain forward-looking information that involves risks and uncertainties, including statements about the use of Hologic's NovaSure system. There can be no assurance this product will achieve the benefits described herein or that such benefits will be replicated in any particular manner with respect to an individual patient. The actual effect of the use of the product can only be determined on a case-by-case basis depending on the particular circumstances and patient in question. In addition, there can be no assurance that this product will be commercially successful or achieve any expected level of sales. Hologic expressly disclaims any obligation or undertaking to release publicly any updates or revisions to any such statements presented herein to reflect any change in expectations or any change in events, conditions or circumstances on which any such statements are based.
This information is not intended as a product solicitation or promotion where such activities are prohibited. For specific information on what products are available for sale in a particular country, please contact a local Hologic sales representative or write to womenshealth@hologic.com.
Hologic, AccuSheath, EndoForm, NovaSure, SureClear and The Science of Sure are trademarks and/or registered trademarks of Hologic Inc. and/or its subsidiaries in the United States and/or other countries.
Notes and Disclaimers
IMPORTANT SAFETY INFORMATION: NovaSure endometrial ablation is for premenopausal women with heavy periods due to benign causes who are finished childbearing. Pregnancy following the NovaSure procedure can be dangerous. The NovaSure procedure is not for those who have or suspect uterine cancer; have an active genital, urinary or pelvic infection; or have an IUD. NovaSure endometrial ablation is not a sterilization procedure. Rare but serious risks include, but are not limited to, thermal injury, perforation and infection. Temporary side effects may include cramping, nausea, vomiting, discharge and spotting. Inform patients to contact you if they experience a possible side effect related to use of this product. For detailed benefit and risk information, please visit www.gynsurgicalsolutions.co.uk/NovaSureV5.
SOURCE: Hologic Inc.
References
1 NovaSure V5 cervical seal drawing, FAB-18821
2 Baggish, MS, Karram, MM. Atlas of Pelvic Anatomy and Gynecologic Surgery. 4th edition. Elsevier 2016. ISBN: 978-0323225526. Chapter 44, Page 493
3 Luo, J et al. Quantitative analyses of variability in normal vaginal shape and dimension on MR images. Int. Urogynecol (2016) 27:1087-1095
4 NovaSure V5 Designs Verification Results, VER-10513
5 NovaSure V5 Instructions for Use, MAN-07653-001
6 Baskett TF, Clough H, Scott TA. NovaSure bipolar radiofrequency endometrial ablation: report of 200 cases. Journal of Obstetricians and Gynecologists Canada. 2005;27(5):473–476.
7 Herman M, Penninx J, Mol B, Bongers M. Ten-year follow-up of a randomised controlled trial comparing bipolar endometrial ablation with balloon ablation for heavy menstrual bleeding. BJOG 2013;120:966–970