Lenzilumab Treatment May Provide Enhanced Likelihood of Survival Without Ventilation in Hospitalized Black and African-American COVID-19 Patients
Lenzilumab Treatment May Provide Enhanced Likelihood of Survival Without Ventilation in Hospitalized Black and African-American COVID-19 Patients
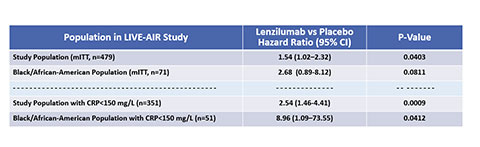
- Lenzilumab treatment in Black and African-American patients in the positive LIVE-AIR Phase 3 trial, having a CRP<150 mg/L, resulted in a nearly 9-fold increase in survival without ventilation
- CDC data shows Black and African-American persons are at an almost 3-fold greater risk of hospitalization and 2-fold greater risk of death from COVID-19 infection1
- Black American vaccination rates are lowest nationally, resulting in higher risk of infection, hospitalization, and death2
BURLINGAME, Calif.--(BUSINESS WIRE)--Humanigen, Inc. (Nasdaq: HGEN) (“Humanigen”), a clinical-stage biopharmaceutical company focused on preventing and treating an immune hyper-response called ‘cytokine storm’, announced analysis of results from its Phase 3 LIVE-AIR study of lenzilumab in hospitalized patients with COVID-19 suggesting Black and African-American patients having a CRP<150 mg/L may be the highest responders to treatment, with a nearly 9-fold increase in likelihood of survival without ventilation (SWOV) [n=51, p-value=0.0412]. In the overall population with CRP<150 mg/L, LIVE-AIR Phase 3 results show patients treated with lenzilumab demonstrated a 2.5-fold increased likelihood of SWOV [mITT, n=351, p-value=0.0009].
“Humanigen believes this analysis is an important finding because Black and African-American patients are hyper-vulnerable to COVID-19,” said Cameron Durrant, MD, Chief Executive Officer, Humanigen. “In light of the rapid ongoing spread of the Delta variant, data suggesting that Black and African-American patients, who are hyper-vulnerable to COVID-19, and may be hyper-responsive to lenzilumab is important in the broader context of the potential benefits that may result if the FDA were to grant emergency use authorization.”
The Centers for Disease Control and Prevention (CDC) has found that race and ethnicity are risk markers for other conditions that affect health, including socioeconomic status, access to health care, and exposure to SARS-CoV-2 related to occupation, such as frontline, essential, and critical infrastructure workers.1 The American Heart Association’s COVID-19 Cardiovascular Disease Registry found that Black patients had the highest prevalence of obesity, hypertension, and diabetes, all of which are medical conditions the CDC identifies as making adults of any age more likely to get severely ill from COVID-19.3,4
“In the interest of public health and safety, it is our priority to share data with stakeholders to improve our understanding of the disease and potential treatments,” said Adrian Kilcoyne, MD, Chief Medical Officer, Humanigen. “While there may be some limitations to subset analyses, we believe the ongoing public health crisis caused by SARS-CoV-2 warrants consideration of these important data by regulatory authorities.”
Humanigen intends to submit data from this analysis for publication in a peer-reviewed journal and present the findings at a medical meeting. These new data from the LIVE-AIR study will also be shared with regulatory authorities in the US, UK, European Union, and other geographies.
About the LIVE-AIR, Phase 3 Study of Lenzilumab
LIVE-AIR Phase 3 study met its primary endpoint of survival without ventilation demonstrating a 1.54-fold improvement overall and trended to a 2.68-fold improvement in Black and African-American patients. This study was a randomized, double-blind, placebo-controlled, multi-center Phase 3 trial for the treatment and prevention of serious and potentially fatal outcomes in patients hospitalized with COVID-19 pneumonia. The primary objective was to assess whether lenzilumab, in addition to other treatments, which included dexamethasone (or other steroids) and/or remdesivir, could alleviate the immune-mediated ‘cytokine storm’ and improve survival without ventilation, or ‘SWOV’ (sometimes referred to as ‘ventilator-free survival’). SWOV is a composite endpoint of time to death and time to invasive mechanical ventilation (IMV), which is a robust measure that is less prone to favor a treatment with discordant effects on survival or days free of ventilation.5
The LIVE-AIR study enrolled 520 patients in 29 sites in the US and Brazil who were at least 18 years of age; experienced blood oxygen saturation (SpO2) of less than or equal to 94%; or required low-flow supplemental oxygen, or high-flow oxygen support, or non-invasive positive pressure ventilation; and were hospitalized but did not require IMV. Following enrollment, subjects were randomized to receive three infusions of either lenzilumab or placebo, each infusion separated by eight hours over a 24-hour period. The primary endpoint was the difference between lenzilumab treatment and placebo treatment in SWOV through day 28 following treatment. Key secondary endpoints, also measured through day 28, included ventilator-free days, duration of ICU stay, incidence of IMV, extracorporeal membrane oxygenation (ECMO), and/or death, time to death, all-cause mortality, and time to recovery. Results of the trial have been submitted for publication in a peer-reviewed journal.
About Humanigen, Inc.
Humanigen, Inc. (Nasdaq: HGEN) (“Humanigen”), a clinical-stage biopharmaceutical company focused on preventing and treating an immune hyper-response called ‘cytokine storm’. Lenzilumab is a first-in class antibody that binds to and neutralizes granulocyte-macrophage colony-stimulating factor (GM-CSF). Results from preclinical models indicate GM-CSF is an upstream regulator of many inflammatory cytokines and chemokines involved in the cytokine storm. Early in the COVID-19 pandemic, investigation showed high levels of GM-CSF secreting T cells were associated with disease severity and intensive care unit admission. Humanigen’s Phase 3 LIVE-AIR study suggests early intervention with lenzilumab may prevent consequences of a full-blown cytokine storm in hospitalized patients with COVID-19. Humanigen is developing lenzilumab as a treatment for cytokine storm associated with CD19-targeted CAR-T cell therapies and exploring the effectiveness of lenzilumab in other inflammatory conditions such as acute Graft versus Host Disease (aGvHD) in patients undergoing allogeneic hematopoietic stem cell transplantation (HSCT), eosinophilic asthma, and rheumatoid arthritis. Humanigen is also developing a portfolio of clinical and pre-clinical therapies for the treatment of inflammation and immuno-oncology. For more information, visit www.humanigen.com and follow Humanigen on LinkedIn, Twitter, and Facebook.
Humanigen Forward-Looking Statements
All statements other than statements of historical facts contained in this press release are forward-looking statements. Forward-looking statements reflect management's current knowledge, assumptions, judgment, and expectations regarding future performance or events. Although management believes that the expectations reflected in such statements are reasonable, they give no assurance that such expectations will prove to be correct, and you should be aware that actual events or results may differ materially from those contained in the forward-looking statements. Words such as "will," "expect," "intend," "plan," "potential," "possible," "goals," "accelerate," "continue," and similar expressions identify forward-looking statements, including, without limitation, statements regarding the effectiveness of lenzilumab in Black and African-American patients; the review of our submission for emergency use authorization by the FDA; and our other plans to explore the effectiveness of lenzilumab and other candidates in our development portfolio as therapies for other inflammation and immune-oncology indications.
Forward-looking statements are subject to a number of risks and uncertainties including, but not limited to, the risks inherent in our lack of profitability and need for additional capital to grow our business; our dependence on partners to further the development of our product candidates; the uncertainties inherent in the development, attainment of the requisite regulatory authorizations and approvals and launch of any new pharmaceutical product; the outcome of pending or future litigation; and the various risks and uncertainties described in the "Risk Factors" sections of our latest annual and quarterly reports and other filings with the SEC.
All forward-looking statements are expressly qualified in their entirety by this cautionary notice. You should not rely upon any forward-looking statements as predictions of future events. The Company undertakes no obligation to revise or update any forward-looking statements made in this filing to reflect events or circumstances after the date hereof or to reflect new information or the occurrence of unanticipated events, except as required by law.
Sources:
- Centers for Disease Control and Prevention. (2021, July 16). Risk for Covid-19 infection, hospitalization, and death by race/ethnicity. Centers for Disease Control and Prevention. https://www.cdc.gov/coronavirus/2019-ncov/covid-data/investigations-discovery/hospitalization-death-by-race-ethnicity.html#print.
- Ndugga, N., Hill, L., Artiga, S., & Parker, N. (2021, July 21). Latest Data On COVID-19 Vaccinations by Race/Ethnicity. Coronavirus (COVID-19). https://www.kff.org/coronavirus-covid-19/issue-brief/latest-data-on-covid-19-vaccinations-race-ethnicity/.
- Rodriguez, F., Solomon, N., de Lemos, J. A., et al. (2020). Racial and Ethnic Differences in Presentation and Outcomes for Patients Hospitalized with COVID-19: Findings from the American Heart Association’s COVID-19 Cardiovascular Disease Registry. Circulation, 143(24), 2332–2342. https://doi.org/10.1161/circulationaha.120.052278
- Centers for Disease Control and Prevention. (2021, May 13). Certain medical conditions and risk for severe covid-19 illness. Centers for Disease Control and Prevention. https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/people-with-medical-conditions.html.
- Novack V., Beitler J., Yitshak-Sade M., et al. (2020). Alive and Ventilator-Free: A Hierarchical, Composite Outcome for Clinical Trials in the Acute Respiratory Distress Syndrome. Critical Care Medicine, 48(2), 158–166. https://doi.org/10.1097/ccm.0000000000004104
Contacts
Humanigen Media
Grace Catlett
RXMD
gcatlett@rxmedyn.com
516-318-8563
Humanigen Investor Relations
Ken Trbovich
Humanigen
trbo@humanigen.com
650-410-3206