BOXBOROUGH, Mass.--(BUSINESS WIRE)--Sterilis LLC, an innovator in regulated medical waste management, today emerged from stealth mode and introduced a dramatically different approach to treating regulated medical waste, enabling healthcare and public health facilities to reduce risk, liability and operational costs, while significantly improving patient and worker safety.
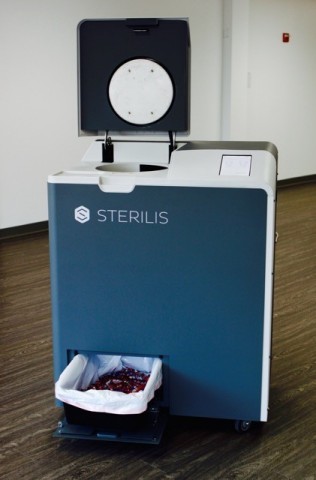
The Sterilis device is a proven alternative to the decades-old method of hauling dangerous waste by truck to be incinerated. Sterilis’ device uses steam sterilization and grinding technology to transform medical waste into a harmless confetti-like material, which can be disposed of in the general waste stream.
Sterilis is poised to disrupt the regulated medical waste (RMW) industry – a $21 billion market globally in 2016, and projected to grow to $33 billion in 2025, as valuated by Grand View Research – for two key reasons. First, healthcare has become increasingly decentralized, with care moving from hospitals to ambulatory or outpatient facilities. Many facilities have limited space, therefore, cost containment is essential and the handling and storage of medical waste is a growing concern. Secondly, medical waste transport costs have continually risen in recent years due to labor costs, fuel prices and waste volume increases. Healthcare administrators have had no choice but to pay continuously high RMW disposal costs. The Sterilis device is a fixed-cost solution that can solve these challenges and help facilities better control expenses.
“The regulated medical waste industry has seen little innovation and still operates under an archaic approach of ‘haul and burn.’ Therefore, the industry is ripe for disruption with Sterilis’ new, safer, and more sustainable approach to treat and safely dispose of regulated medical waste, when and where it is generated,” said Bob Winskowicz, CEO of Sterilis. “The Sterilis device makes handling medical waste safer, protects healthcare professionals, reduces costs, time and labor, and reduces the need to incinerate medical waste. We’re excited by the response and customer demand for the Sterilis device as we enter a more aggressive growth phase and expand our sales and distribution footprint nationwide.”
Sterilis is already realizing significant demand for its device in a variety of healthcare segments. Devices are in use at hospitals, dialysis clinics, surgery centers, dermatology and ophthalmology practices, needle drop-off centers, health clinics, and clinical labs.
Sterilis is also seeing demand for its device in local needle exchange facilities, which are challenged with reducing needle-related infections in their communities, as the nationwide opioid crisis has led to an overwhelming increase in the number of needles communities must dispose of safely. Many needles are dispensed at a local pharmacy or needle exchange, but some self-injectors, without a suitable method of disposal, are throwing needles away in the regular garbage while others throw them on the ground. The Sterilis device provides a way for communities to safely dispose of needles and eliminate the problem immediately.
Additionally, demand for Sterilis’ device is coming from facilities looking to address their emergency preparedness plans for situations such as natural disasters, disease outbreaks, or terrorism. The device is a portable, self-contained unit providing on-site remediation of RMW for quarantined areas when conventional methods of disposal are incapacitated.
Sterilis’ device uses a patented design that combines a steam sterilizer with a grinder and allows RMW to be treated on-site and on-demand at point-of-care in about 60 minutes. As a result of grinding the waste, the device reduces the volume of waste by 80 percent, and the sterilized confetti can be thrown out in the regular trash. It includes software that tracks all regulatory data and stores it in the cloud, where it can be accessed and monitored at any time.
“The Sterilis device gives our organization two significant benefits: waste reduction and cost savings,” said Barbara Plansinis, Administrator at Wentworth Surgery Center. “Previously, we were boxing and shipping out waste by the ton. By comparison, the amount of waste we are disposing of now, using the Sterilis device, is minimal. In addition, we’ve cut our waste remediation costs by about 25 percent.”
Sterilis has assembled a world-class advisory board comprised of respected healthcare and medical leaders. These include leading physicians, medical executives, prestigious medical professors and business consultants. Building this team of advisors, along with its sales team and strategic distribution partnerships, has put Sterilis in strong position for growth in 2017.
For more information about the Sterilis solution, visit www.sterilismedical.com, or follow Sterilis on Twitter, @sterilis_llc.
About Sterilis
Sterilis LLC has developed a patented
technology that converts regulated medical waste into harmless,
confetti-like material that can be thrown out with the regular trash.
Sterilis devices are American-made in Springfield, Massachusetts and are
in use at customer sites in healthcare, public safety (fire and police
departments) and aviation. Sterilis devices provide ease of use, safety,
convenience, environmental sustainability and cost saving benefits, all
in one device. The company is privately-held and headquartered in
Boxborough, Mass.