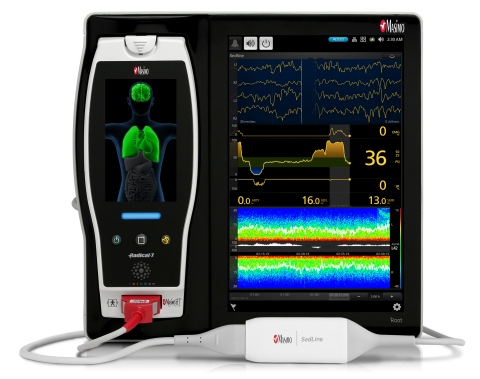
NEUCHATEL, Switzerland--(BUSINESS WIRE)--Masimo (NASDAQ: MASI) announced today the CE mark and scheduled full market release of Next Generation SedLine® Brain Function Monitoring technology at the European Society of Anaesthesiology’s (ESA) 2016 Euroanaesthesia Congress in London, England.
Next Generation SedLine enhances how Masimo’s processed EEG parameter, the Patient State Index (PSI), responds in challenging situations, addressing many of the concerns raised with quantitative EEG while bolstering brain function monitoring’s support of anesthetic management.
Next Generation PSI uses Masimo’s breakthrough Parallel Engines and Adaptive Signal Processing technology and provides the following enhancements:
- Less susceptibility to electromyography (EMG) interference by extracting a clearer EEG signal even in the presence of EMG
- Improved PSI performance in low power EEG cases
These improvements build upon the existing benefits of SedLine technology:
- Four simultaneous EEG leads to enable continuous assessment of both sides of the brain
- Density Spectral Array (DSA) offers easy-to-interpret, high-resolution display of bi-hemispheric activity
- Multiple screen views expand information while enabling customization in the OR and ICU
- Electrocautery resistance
“We are delighted to unveil Next Generation SedLine,” said Joe Kiani, Founder and CEO of Masimo. “Next Generation PSI takes advantage of Masimo’s signal processing prowess and promises to do for brain function monitoring what SET did for pulse oximetry.”
Next Generation SedLine has not received FDA 510(k) clearance and is not currently available for sale in the United States.
@MasimoInnovates | #Masimo
About Masimo
Masimo (NASDAQ: MASI) is a global leader in innovative noninvasive monitoring technologies. Our mission is to improve patient outcomes and reduce the cost of care by taking noninvasive monitoring to new sites and applications. In 1995, the company debuted Masimo SET® Measure-through Motion and Low Perfusion™ pulse oximetry, which has been shown in multiple studies to significantly reduce false alarms and accurately monitor for true alarms. Masimo SET® is estimated to be used on more than 100 million patients in leading hospitals and other healthcare settings around the world. In 2005, Masimo introduced rainbow® Pulse CO-Oximetry technology, allowing noninvasive and continuous monitoring of blood constituents that previously could only be measured invasively, including total hemoglobin (SpHb®), oxygen content (SpOC™), carboxyhemoglobin (SpCO®), methemoglobin (SpMet®), and more recently, Pleth Variability Index (PVI®) and Oxygen Reserve Index (ORI™), in addition to SpO2, pulse rate, and perfusion index (PI). In 2014, Masimo introduced Root®, an intuitive patient monitoring and connectivity platform with the Masimo Open Connect (MOC-9) interface. Masimo is also taking an active leadership role in mHealth with products such as the Radius-7™ wearable patient monitor and the MightySat™ fingertip pulse oximeter. Additional information about Masimo and its products may be found at www.masimo.com.
Forward-Looking Statements
This press release includes forward-looking statements as defined in Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934, in connection with the Private Securities Litigation Reform Act of 1995. These forward-looking statements include, among others, statements regarding the potential effectiveness of Masimo SedLine®. These forward-looking statements are based on current expectations about future events affecting us and are subject to risks and uncertainties, all of which are difficult to predict and many of which are beyond our control and could cause our actual results to differ materially and adversely from those expressed in our forward-looking statements as a result of various risk factors, including, but not limited to: risks related to our assumptions regarding the repeatability of clinical results; risks related to our belief that Masimo's unique noninvasive measurement technologies, including Masimo SedLine®, contribute to positive clinical outcomes and patient safety; risks related to our belief that Masimo noninvasive medical breakthroughs provide cost-effective solutions with comparable accuracy and unique advantages, including: immediate and continuous results that enable earlier treatment without causing invasive trauma in all patients and in every clinical situation; as well as other factors discussed in the "Risk Factors" section of our most recent reports filed with the Securities and Exchange Commission ("SEC"), which may be obtained for free at the SEC's website at www.sec.gov. Although we believe that the expectations reflected in our forward-looking statements are reasonable, we do not know whether our expectations will prove correct. All forward-looking statements included in this press release are expressly qualified in their entirety by the foregoing cautionary statements. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as of today's date. We do not undertake any obligation to update, amend or clarify these statements or the "Risk Factors" contained in our most recent reports filed with the SEC, whether as a result of new information, future events or otherwise, except as may be required under the applicable securities laws.