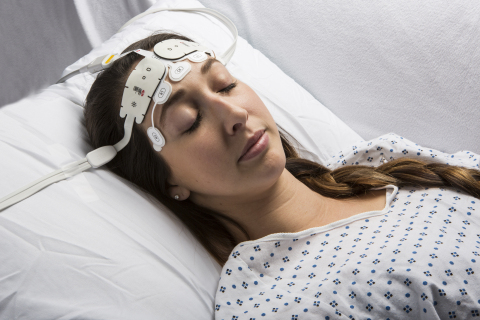
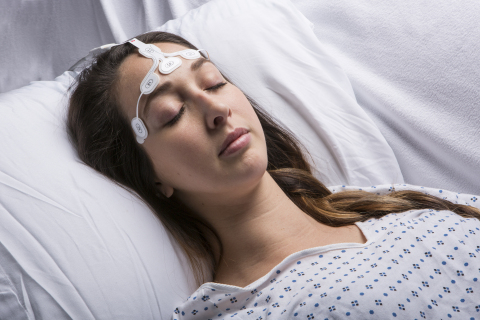
IRVINE, Calif.--(BUSINESS WIRE)--Masimo (NASDAQ: MASI) announced today availability of the RD SedLine® EEG sensor, for use with Masimo SedLine Brain Function Monitoring and compatible with simultaneous use of Masimo O3™ Regional Oximetry. SedLine and O3 provide simultaneous monitoring on the Masimo Root® monitoring platform, helping to give clinicians more information about the brain.
The RD SedLine EEG sensor features a repositioned, color-coded sensor-cable connection that lies comfortably on the patient’s head and soft foam pads to reduce discomfort upon application to the patient. The sensor’s streamlined shape and built-in fitting guide allow simultaneous application of SedLine and O3 sensors. The sensor’s performance and specifications remain the same and work with existing SedLine modules, via an updated patient cable.
SedLine brain function monitoring features four simultaneous EEG leads to enable continuous assessment of both sides of the brain, four EEG waveforms, a Density Spectral Array (DSA; an easy-to-interpret, high-resolution display of bi-hemispheric activity and EEG power), and the Patient State Index (PSI; a processed EEG parameter related to the effect of anesthetic agents). Next Generation SedLine, available outside the U.S., enhances the PSI to make it less susceptible to electromyographic (EMG) interference and to improve performance in low-power EEG cases.
O3 regional oximetry uses near-infrared spectroscopy (NIRS) to continuously monitor absolute and trended regional tissue oxygen saturation (rSO2) in the cerebral region. Regional oximetry may help clinicians monitor cerebral oxygenation in situations in which pulse oximetry alone may not be fully indicative of the oxygen in the brain due to various factors, such as the type of clinical procedure being performed.
Dr. David Drover, Professor of Anesthesiology in the Department of Anesthesiology, Perioperative and Pain Medicine at Stanford Hospital, stated, “The RD SedLine sensor allows simultaneous application with O3 Regional Oximetry to deliver more information about my patient’s brain in a single specialty monitor.”
“Root with SedLine and O3 presents a powerful brain monitoring solution,” said Joe Kiani, Founder and CEO of Masimo. “With the addition of the RD SedLine EEG sensor, the EEG and optical sensors fit together like puzzle pieces, making it easier for clinicians to simultaneously monitor patients with both technologies, while providing a comfortable experience for the patient.”
The RD SedLine EEG sensor is available in the U.S. Next Generation SedLine does not have 510(k) clearance and is not available in the U.S.
@MasimoInnovates | #Masimo
About Masimo
Masimo (NASDAQ: MASI) is a global leader in innovative noninvasive monitoring technologies. Our mission is to improve patient outcomes and reduce the cost of care by taking noninvasive monitoring to new sites and applications. In 1995, the company debuted Masimo SET® Measure-through Motion and Low Perfusion™ pulse oximetry, which has been shown in multiple studies to significantly reduce false alarms and accurately monitor for true alarms. Masimo SET® has also been shown to help clinicians reduce severe retinopathy of prematurity in neonates,1 improve CCHD screening in newborns,2 and, when used for continuous monitoring in post-surgical wards, reduce rapid response activations and costs.3,4,5 Masimo SET® is estimated to be used on more than 100 million patients in leading hospitals and other healthcare settings around the world. In 2005, Masimo introduced rainbow® Pulse CO-Oximetry technology, allowing noninvasive and continuous monitoring of blood constituents that previously could only be measured invasively, including total hemoglobin (SpHb®), oxygen content (SpOC™), carboxyhemoglobin (SpCO®), methemoglobin (SpMet®), and more recently, Pleth Variability Index (PVi®) and Oxygen Reserve Index (ORi™), in addition to SpO2, pulse rate, and perfusion index (PI). Studies with SpHb have shown reductions in unnecessary blood transfusion*,6,7 and when used with PVi, reductions in length of hospital stay8 and 30- and 90-day mortality.9 In 2014, Masimo introduced Root®, an intuitive patient monitoring and connectivity platform with the Masimo Open Connect™ (MOC-9™) interface, enabling other companies to augment Root with new features and measurement capabilities. Masimo is also taking an active leadership role in mHealth with products such as the Radius-7™ wearable patient monitor, iSpO2® pulse oximeter for smartphones, and the MightySat™ fingertip pulse oximeter. Additional information about Masimo and its products may be found at www.masimo.com. Published clinical studies on Masimo products can be found at http://www.masimo.com/cpub/clinical-evidence.htm.
*Clinical decisions regarding red blood cell transfusions should be based on the clinician’s judgment considering, among other factors: patient condition, continuous SpHb monitoring, and laboratory diagnostic tests using blood samples.
References
- Castillo A et al. Prevention of Retinopathy of Prematurity in Preterm Infants through Changes in Clinical Practice and SpO2 Technology. Acta Paediatr. 2011 Feb;100(2):188-92.
- de-Wahl Granelli A et al. Impact of pulse oximetry screening on the detection of duct dependent congenital heart disease: a Swedish prospective screening study in 39,821 newborns. BMJ. 2009;338.
- Taenzer AH et al. Impact of Pulse Oximetry Surveillance on Rescue Events and Intensive Care Unit Transfers: A Before-And-After Concurrence Study. Anesthesiology. 2010; 112(2):282-287.
- Taenzer AH et al. Postoperative Monitoring – The Dartmouth Experience. Anesthesia Patient Safety Foundation Newsletter. Spring-Summer 2012.
- McGrath SP et al. Surveillance Monitoring Management for General Care Units: Strategy, Design, and Implementation. The Joint Commission Journal on Quality and Patient Safety. 2016 Jul;42(7):293-302.
- Ehrenfeld JM et al. Continuous Non-invasive Hemoglobin Monitoring during Orthopedia Surgery: A Randomized Trial. J Blood Disorders Transf. 2014. 5:9. 2.
- Awada WN et al. Continuous and noninvasive hemoglobin monitoring reduces red blood cell transfusion during neurosurgery: a prospective cohort study. J Clin Monit Comput. 2015 Feb 4.
- Thiele RH et al. Standardization of Care: Impact of an Enhanced Recovery Protocol on Length of Stay, Complications, and Direct Costs after Colorectal Surgery. JACS (2015). doi: 10.1016/j.jamcollsurg.2014.12.042.
- Nathan N et al. Impact of Continuous Perioperative SpHb Monitoring. Proceedings from the 2016 ASA Annual Meeting, Chicago. Abstract #A1103.
Forward-Looking Statements
This press release includes forward-looking statements as defined in Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934, in connection with the Private Securities Litigation Reform Act of 1995. These forward-looking statements include, among others, statements regarding the potential effectiveness of Masimo SedLine®, RD SedLine EEG sensors, and O3™. These forward-looking statements are based on current expectations about future events affecting us and are subject to risks and uncertainties, all of which are difficult to predict and many of which are beyond our control and could cause our actual results to differ materially and adversely from those expressed in our forward-looking statements as a result of various risk factors, including, but not limited to: risks related to our assumptions regarding the repeatability of clinical results; risks related to our belief that Masimo's unique noninvasive measurement technologies, including Masimo SedLine, RD SedLine EEG sensors, and O3, contribute to positive clinical outcomes and patient safety; as well as other factors discussed in the "Risk Factors" section of our most recent reports filed with the Securities and Exchange Commission ("SEC"), which may be obtained for free at the SEC's website at www.sec.gov. Although we believe that the expectations reflected in our forward-looking statements are reasonable, we do not know whether our expectations will prove correct. All forward-looking statements included in this press release are expressly qualified in their entirety by the foregoing cautionary statements. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as of today's date. We do not undertake any obligation to update, amend or clarify these statements or the "Risk Factors" contained in our most recent reports filed with the SEC, whether as a result of new information, future events or otherwise, except as may be required under the applicable securities laws.